In the rapidly evolving world of structural biology, Cryo-Electron Microscopy (cryo-EM) has moved from a “fringe” technique to the gold standard for drug discovery and molecular research. This “resolution revolution” allows scientists to see the building blocks of life at an atomic level without the need for the rigid, often impossible, process of protein crystallization.
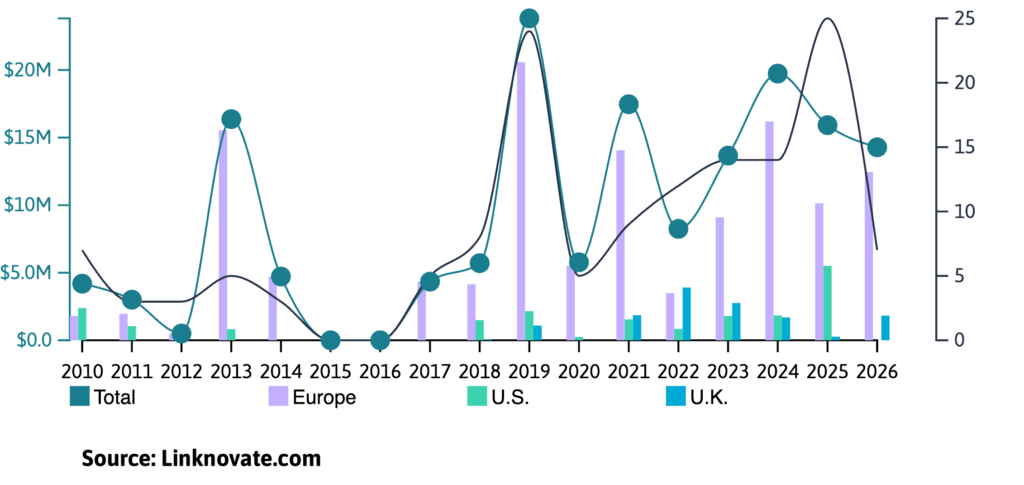
As Linknovate data shows, research and development in cryo-EM has kept a positive growth in the past decade, and 2026 is expected to follow this trend.
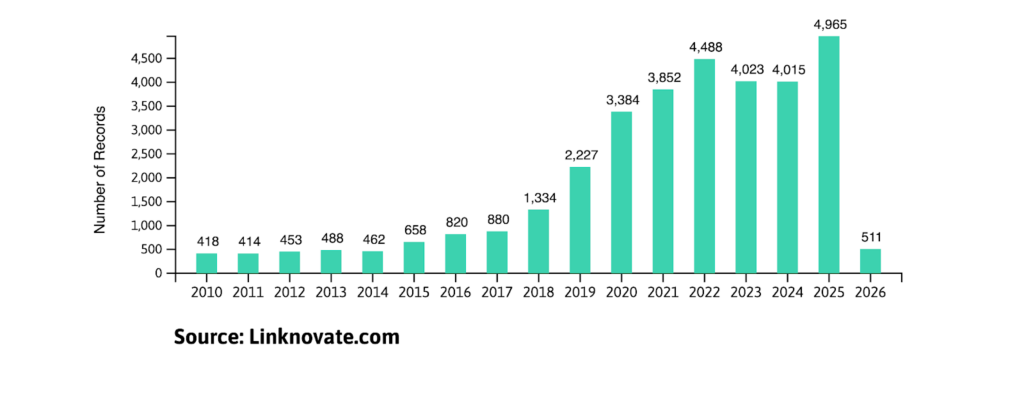
Whether you are a biotech investor, a researcher, or a pharmaceutical lead, understanding the current landscape of cryo-EM is essential for staying ahead in 2026.
What is Cryo-EM? The Future of Structural Biology
CryoEM is a transmission electron microscopy technique used to image samples kept at cryogenic temperature. By maintaining the sample at cryogenic temperatures (typically below -180°C), the technique preserves the hydrated structure of proteins, viruses, and complex molecular machines.
Key Benefits Driving the Market:
- Native State Imaging: Unlike X-ray crystallography, cryo-EM allows molecules to remain in a near-native hydrated environment.
- Near-Atomic Resolution: Recent advances in direct electron detectors (DED) now permit resolutions below 1.2 Ångströms.
- Dynamic Analysis: Researchers can capture “snapshots” of proteins in different functional states, revealing the dynamics of how molecules actually work.
Emerging Leaders in Cryo-EM (2026)
While cryo-EM remains rooted in academia (with research publications still outpacing patent filings, and academia leading R&D), a new wave of commercial pioneers is successfully transitioning this technology from the lab to the global market.
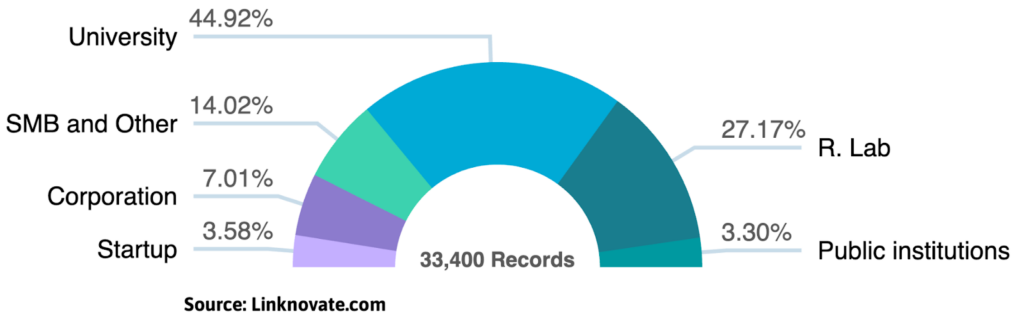
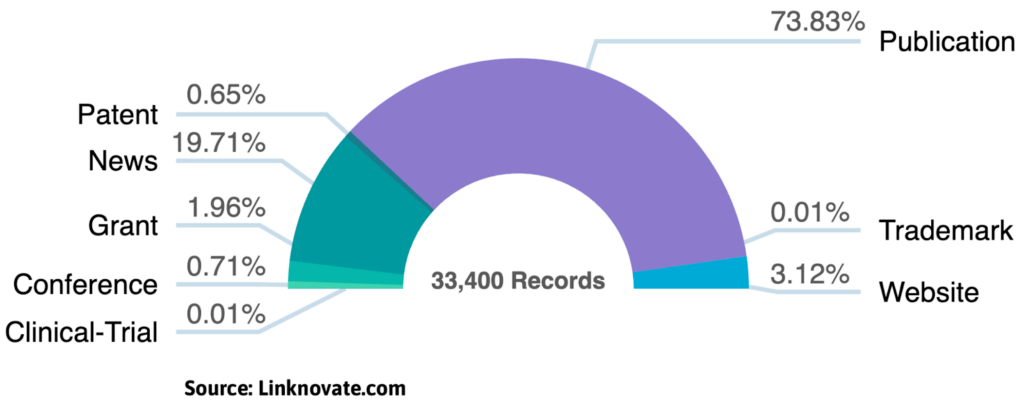
While giants like Thermo Fisher and JEOL dominate the hardware market, a new wave of agile companies and specialized Contract Research Organizations (CROs) are making the technology accessible and efficient.
1. TEMPoS Microscopy (Santiago de Compostela, Spain)
Hailing from the biotech hub of Galicia, TEMPoS is a standout emerging leader specializing in high-end cryo-EM services,offering a dual-track technological approach that addresses the two primary bottlenecks in modern biopharma: Drug Discovery and Drug Delivery.
- Drug Discovery (Structural Biology): They provide a full-stack pipeline—from sample vitrification to 3D model building—focused on high-resolution structure determination of complex targets, such as GPCRs and membrane proteins, at near-atomic scale.
- Drug Delivery (Nanoparticle Analytics): Through their proprietary SaaS platform, TEMPoS offers specialized, automated characterization of Advanced Drug Delivery Systems (ADDS). This digital-first approach provides critical metrics for quality control, payload distribution, and capsid integrity for AAVs, LNPs, and other synthetic vectors.
Expertise: By combining physical lab excellence with a high-throughput analytical platform, they bridge the gap between complex raw data and actionable biological insights for both small molecules and advanced therapies.
Why they matter: TEMPoS is democratizing access to top-tier structural biology. Their model allows startups and academic labs to access state-of-the-art hardware and high-end scientific computing power without the multi-million dollar overhead of housing their own infrastructure.
Specialty: Their dual focus makes them a unique strategic partner: resolving atomic structures for rational drug design while simultaneously accelerating the advanced therapeutics and nanomedicine sectors through their specialized SaaS-led delivery analysis platform.
2. CryoCloud (Utrecht, Netherlands)
Something cool about CryoCloud: they are a young, innovative team transforming the cryo-EM data workflow. While the microscope captures the images, CryoCloud is revolutionizing the data-heavy aftermath. Cryo-EM generates massive datasets (often multiple terabytes) that traditionally required expensive, on-site supercomputers.
CryoCloud solves this with a cloud-native platform optimized for structural biology, allowing researchers to spin up powerful computing clusters on demand. By making high-speed data processing accessible via a web browser, they enable biotechs to reach atomic resolution in days instead of months, eliminating the need for costly in-house server rooms.
3. Gandeeva Therapeutics (Vancouver, Canada)
Gandeeva is bridging the gap between atomic imaging and artificial intelligence. The company has developed a world-leading platform that integrates CRYO-CADD™ and specialized “molecular glue” technology to tackle previously “undruggable” targets in oncology and infectious diseases. Their innovative approach, highlighted by high-stakes collaborations with giants like Moderna, transforms cryo-EM from a static imaging tool into a dynamic, AI-driven engine capable of discovering life-saving therapeutics with unprecedented speed and structural precision.
4. CryoSol and CRyoWrite
CryoSol and CryoWrite are automating sample preparation, the field’s most notorious bottleneck. By replacing inconsistent manual “blotting” with precision technologies like VitroJet’s pin-printing (CryoSol) and nanoliter microcapillary writing (cryoWrite), these companies ensure perfectly thin, reproducible ice layers while using minimal protein volumes. For a data-driven company, this shift is critical: it transforms a high-failure craft into a standardized, high-throughput robotic process, providing the pristine, high-quality images necessary for advanced AI structural analysis.
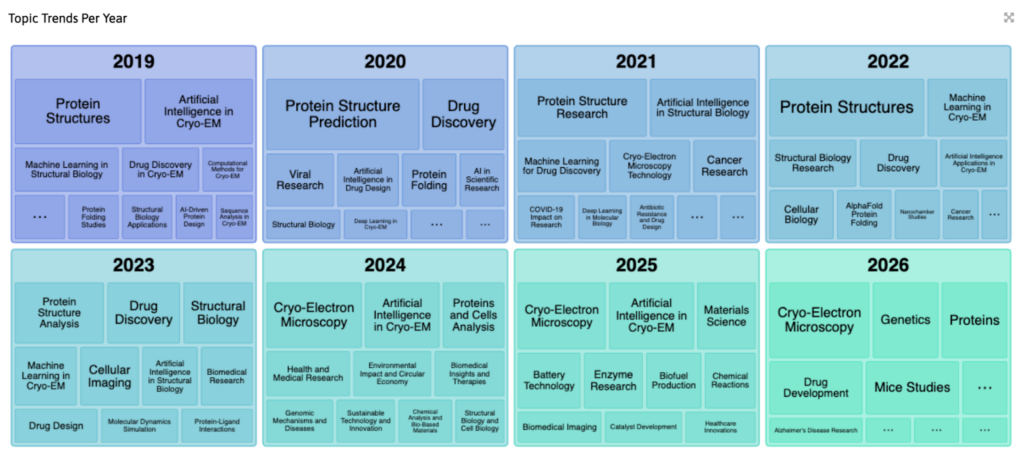
Cryo-EM Trends to Watch in 2026
The industry is currently shifting from discovery to throughput. Here is what’s trending:
AI-Driven CryoEM Workflow
Software like Relion and CryoSPARC are being integrated with AI to automate particle picking, classification, 3D reconstruction, segmentation, practically reducing data processing time from weeks to hours.
AlphaFold/ML models + Cryo‑EM synergy
AI-based structure prediction is now routinely used to guide model building, interpret ambiguous densities, and integrate predicted conformational ensembles into experimental maps, making structure determination faster and more accurate.
In-situ Cryo-ET
Scientists are no longer just looking at isolated proteins; they are using Focus Ion Beam (FIB) milling combined with Cryo-Electron Tomography to look inside entire cells to see how proteins interact in their “crowded” natural habitat.
Higher throughput and automation
Modern Cryo‑EM microscopes and software enable multi-sample runs, remote operation, and automated queuing, increasing data output and allowing researchers to screen large numbers of samples efficiently.
Membrane protein and complex stability advancements
Innovations in detergents, nanodiscs, and other stabilizing agents improve the yield and structural quality of challenging targets such as GPCRs and large multiprotein assemblies.
Cryo‑EM for nanoparticles and gene therapy vectors
Cryo‑EM is becoming a key tool for characterizing viral vectors, exosomes, and lipid nanoparticles, providing insights into capsid integrity, particle heterogeneity, and aggregation that are essential for drug delivery and regulatory purposes.
Democratization of EM
Access to Cryo‑EM is expanding through lower-cost instruments, shared facilities, and cloud-based analysis, allowing smaller labs and new researchers to perform high-quality structural studies without owning expensive equipment.
Why Invest in Cryo-EM Services Now?
For pharmaceutical companies, the ROI is clear. Understanding the exact binding pocket of a drug candidate can shave years off the development cycle.
Beyond classical small-molecule targets, Cryo‑EM is now proving indispensable for characterizing nanoparticles and gene therapy vectors like AAVs, LNPs, and exosomes. Detailed structural insights allow companies to assess particle integrity, heterogeneity, and aggregation, critical parameters for safe and effective drug delivery.
As companies like TEMPoS continue to refine the service-based model, even small-scale biotechs can now leverage Nobel Prize-winning technology to develop the next generation of life-saving therapeutics.
Stay ahead of the Cryo-EM Ecosystem
The world of cryo-EM moves fast. With new breakthroughs in resolution and software emerging at a rapid pace, manually tracking the shift from academic papers to industrial patents is a full-time job. To truly lead in this space, you need more than just a Google search; you need competitive intelligence.
Scout the Future with Linknovate
If you want to identify the next TEMPoS or track the latest patent filings before they hit the mainstream news, try Linknovate.
Linknovate is an AI-powered innovation scouting platform designed to help R&D teams and strategy leads navigate the complex landscape of deep tech. By aggregating data from scientific publications, patents, grants, and news, Linknovate allows you to:
- Identify Hidden Players: Discover emerging startups and agile SMEs in structural biology that are flying under the radar.
- Track R&D Trends: Monitor the “weak signals” of technology, seeing where the money and research are moving before the market catches up.
- Benchmark Competitors: Keep a pulse on what other biotech and pharma giants are investigating in real-time.
Don’t just watch the field grow: scout your next partnership or breakthrough with precision.