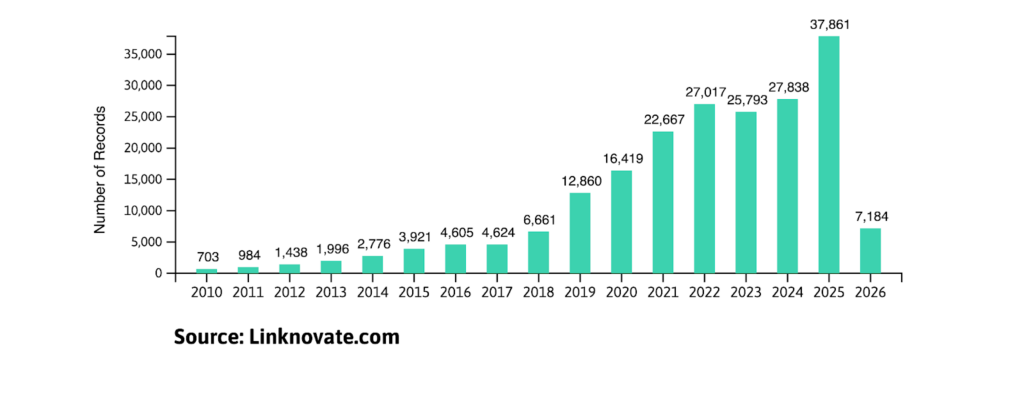
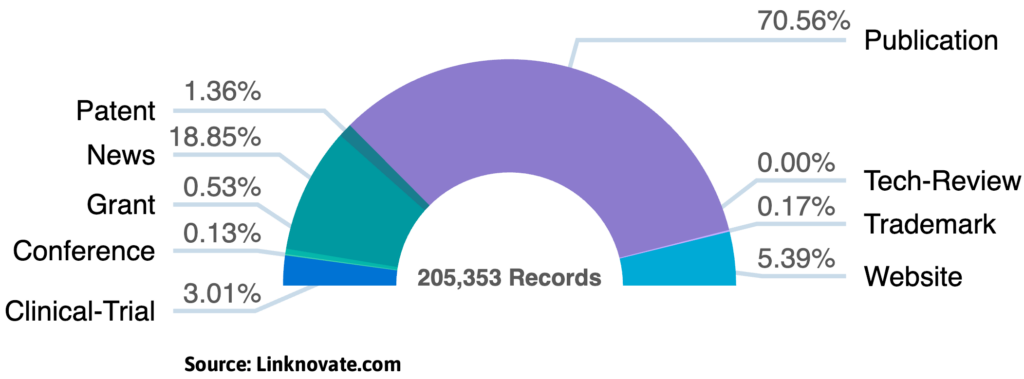
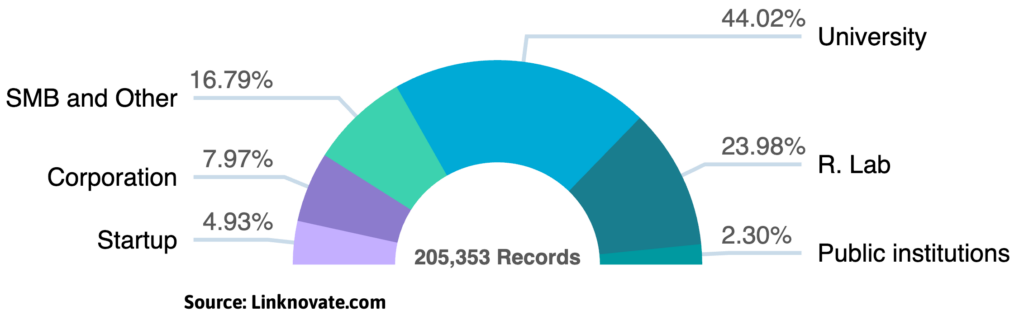
The Microbiome-based Therapeutics R&D sector is projected to maintain its double-digit growth throughout 2026, following a transformative “boom” year in 2025. With the human microbiome market size now exceeding $1.6 billion, the focus has shifted toward targeted microbial strains and AI-driven drug discovery.
Using Linknovate’s proprietary data, including biotech patent analytics, Venture Capital (VC) trends, and clinical trial milestones, we’ve mapped the most influential companies in the space.
Key Insights for 2026:
- Academic Leadership: University-led research remains the primary driver of scientific publications in gut-brain axis and metabolic health.
- Niche Market Growth: Agile startups and SMBs are successfully outpacing legacy firms in securing innovative patents for personalized medicine.
- Geographic Hubs: R&D activity is heavily concentrated in the US (Cambridge, NYC) and Europe (Paris, London, Madrid).
Ready to see who is leading the next wave of microbial innovation? Here are the Top 10 Microbiome Therapeutics startups to watch this year.
1. Mikrobiomik (Spain)
This Bilbao-based startup is set to launch Spain’s first authorized human microbiome therapy, EUTEGRA (MBK-01), in 2026.
- Target: Recurrent Clostridioides difficile infection.
- Innovation: They have successfully standardized fecal microbiota treatments into an oral capsule format under strict GMP (Good Manufacturing Practice) standards.
2. MaaT Pharma (France)
MaaT Pharma is leading the charge in Microbiome-Oncology. They focus on restoring gut-immune homeostasis in patients undergoing cancer treatment.
- The Breakthrough: Their lead candidate, Xervyteg (MaaT013), recently showed a three-fold increase in one-year survival for patients with Graft-versus-Host Disease (GvHD).
- Status: Currently seeking EMA marketing authorization in 2026.
3. Vedanta Biosciences (USA)
A pioneer in “defined bacterial consortia,” Vedanta doesn’t just transplant stool; they manufacture precise, lab-grown “pill-based” medicines.
- The 2026 Milestone: All eyes are on the topline data from their Phase 3 RESTORATiVE-303 study for recurrent C. difficile.
- Why it matters: Unlike fecal transplants, Vedanta’s approach offers a standardized, scalable drug product that can be prescribed like any traditional pharmaceutical.
4. Eligo Bioscience (France)
Eligo is moving beyond “adding” bacteria to “editing” them. They use CRISPR-based technology to target and eliminate specific harmful bacterial strains while leaving the beneficial ones intact.
- Focus: Immuno-dermatology and genetic diseases.
- Funding: Following a $30M Series B, they are advancing their EB005 candidate for inflammatory skin conditions.
5. Seres Therapeutics (USA)
As the creators of the first-ever FDA-approved oral microbiome drug (VOWST), Seres remains a heavyweight.
- The 2026 Pipeline: They are now pushing SER-155 through Phase 1b/2 trials to prevent infections in patients receiving stem cell transplants.
- Strategic Edge: Their deep partnership with Nestlé Health Science provides a commercial roadmap that most startups lack.
6. Infant Bacterial Therapeutics – IBT (Sweden)
IBT focuses on one of the most vulnerable populations: premature infants.
- The Candidate: IBP-9414, designed to prevent Necrotizing Enterocolitis (NEC).
- 2026 Update: Potential FDA accelerated approval is on the horizon, which could make this a standard-of-care in neonatal intensive care units globally.
7. Exeliom Biosciences (France)
Exeliom is built around a single “super-bacterium”: Faecalibacterium prausnitzii.
- The Science: This specific strain is known for its potent anti-inflammatory properties.
- Therapeutic Area: They are targeting Crohn’s disease and immuno-oncology, positioning themselves as a leader in “Next-Generation Probiotics” (NGPs).
8. Locus Biosciences (USA)
Locus uses CRISPR-Cas3-engineered bacteriophages to kill specific antibiotic-resistant bacteria.
- Innovation: While most CRISPR tech edits human cells, Locus uses it as a precision-guided missile to destroy bacterial DNA.
- Market Move: Their platform is highly relevant in the global fight against Antimicrobial Resistance (AMR).
9. Metagen Therapeutics (Japan)
Emerging from Japan’s “Drug Discovery Venture Ecosystem,” Metagen is building a massive gut-microbiome drug platform.
- Key Asset: An oral FMT (Fecal Microbiota Transplant) drug candidate for Ulcerative Colitis.
- Strategy: They are leveraging Japan’s unique regulatory environment to accelerate clinical trials for inflammatory bowel disease (IBD).
10. MRM Health (Belgium)
MRM Health recently secured a landmark €55M Series B to advance their Live Biotherapeutic Products (LBPs).
- The Tech: Their CORAL® platform allows for the manufacturing of complex, multi-strain consortia in a single process.
- Clinical Focus: They are currently investigating treatments for Parkinson’s disease and Type 2 Diabetes, highlighting the growing importance of the gut-brain and gut-metabolism axes.
The Future of Microbiome R&D in 2026
The expansion of the microbiome therapeutics market into specialized fields like neuro-inflammation and skin homeostasis marks a definitive turning point for global biotechnology. As we move through 2026, we are witnessing the long-awaited convergence of academic discovery and commercial scalability, where the focus has shifted from simple gut health to the development of high-precision Live Biotherapeutic Products. This diversification into the gut-brain and gut-skin axes is no longer theoretical; it is being fueled by a surge in Phase III clinical trials and a 30% annual growth rate that is drawing heavy investment from both Big Pharma and specialized venture capital.
In this high-velocity environment, the competitive advantage belongs to R&D leaders who can successfully navigate a landscape crowded with CRISPR-edited microbial strains and complex patent portfolios. Success now requires more than just tracking broad trends. It demands the ability to pinpoint emerging startups and specific bacterial consortia before they clear the final regulatory hurdles at the FDA or EMA.
At Linknovate, we empower innovation scouts by mapping the exact players redefining human health, providing the data-driven clarity needed to turn overwhelming clinical and patent data into a decisive strategic edge.